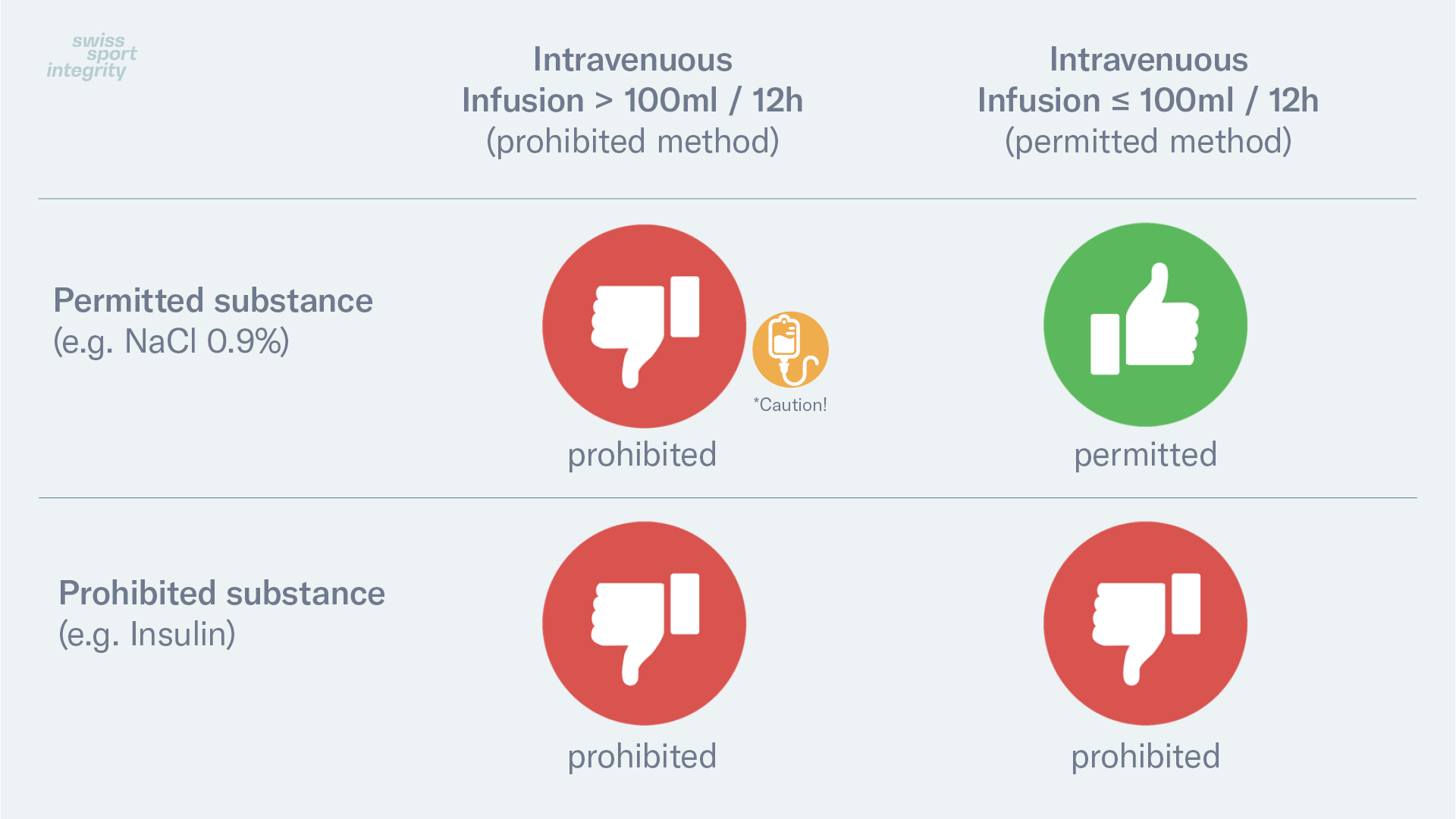
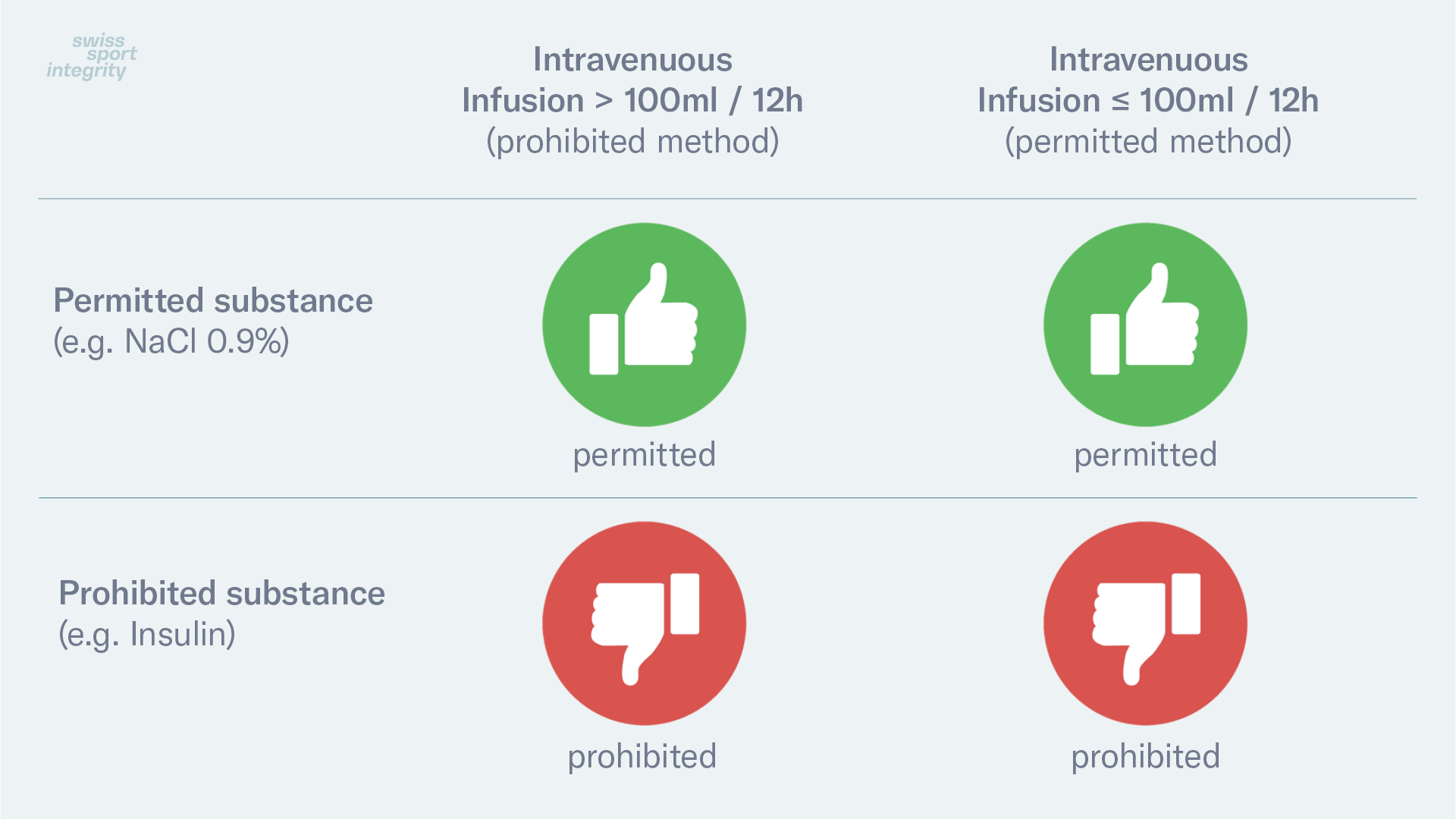
According to the Prohibited List, intravenous infusions and/or injections of more than 100 ml per 12-hour period of any substance are a prohibited method, even if the substance administered is not prohibited. This does not include intravenous infusions and/or injections, which are legitimately received in the course of
- hospital treatments;
- surgical procedures;
- or during clinical diagnostic investigations.
Based on this applicable regulation according to the Prohibited List and of the principle that a substance and/or a method may be prohibited, various possible combinations exist.
Prohibited Status of Intravenous Infusions
*Caution! In the medication inquiry service Global DRO, the prohibited status is display for substances. The information regarding the prohibited intravenous infusions is added after this information as a condition / warning, but it is not displayed graphically (green/red).
If your intravenous infusion is/was prohibited, inquire about TUE applications and medical emergencies.
Practical Example: Intravenous Iron Infusions
The oral intake of an iron product is permitted). However, the above-mentioned regulation applies also to intravenous infusions and/or injections of iron.
This means that intravenous iron infusions of more than 100 ml per 12-hour period are prohibited in accordance with the Prohibited List.
For athletes with an iron deficiency, oral substitution is therefore the therapy of choice. If oral therapy does not have a sufficient effect, parenteral iron ca be administered slowly intravenously by injection (undiluted or diluted with NaCl 0.9% up to 100 ml).
If, for medical reasons, there is a necessity for an intravenous iron infusion with a volume of more than 100 ml, for which none of the above permitted therapy alternatives can be used, a therapeutic use exemption (TUE) is required. The TUE Wizard will show you at what time and to which anti-doping organization the TUE application must be submitted.
In all cases, in addition to the usual medical documentation and diagnosis, a comprehensive medical justification why no permitted therapy alternative can be used is required. If such a justification is missing, inadequate according to the applicable regulations or not comprehensible to the TUE committee, no TUE will be granted.